Lung Cancer Choices© 6th Edition Menu
Chapter 1: Diagnosis and Staging of Lung Cancer
Chapter 2: Comprehensive Biomarker Testing
Chapter 3: Surgery for Lung Cancer Patients
Chapter 5: Radiation Therapy for Non-Small Cell Lung Cancer
Chapter 6: Treatment for Small Cell Lung Cancer
Chapter 7: Clinical Trials and Emerging Therapies for Lung Cancer
Chapter 9: Nutrition in the Patient with Lung Cancer
Chapter 10: Sexuality and Lung Cancer
Chapter 11: Integrative Medicine, Complementary Therapies, and Chinese Medicine in Lung Cancer
Chapter 12: Lung Cancer in People who have Never Smoked
Chapter 13: How to Quit Smoking Confidently and Successfully
Lung Cancer Question Builder - Develop your list of "Questions to Ask" your providers
Treatment of Small Cell Lung Cancer
Ticiana Leal, MD, Selina K. Wong, MD, FRCPC, Join Y. Luh MD, FACP, FACR, and Charles R. Thomas, Jr. MD, FASCO, FASTRO
Introduction
An estimated 238,340 new cases of lung cancer will be diagnosed in the United States in 2023. There are two main types of lung cancer, non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). SCLC accounts for 12 to 15% of all new lung cancer diagnosis.1 It is important that all patients with lung cancer know what type they have because the treatments as well as side effect profiles may differ. Furthermore, treatment for one type may not be effective for another.
This chapter will review the approach to SCLC. See Chapter 4: Systemic Therapy for Non-Small Cell Lung Cancer and Chapter 5: Radiation Therapy for Non-Small Cell Lung Cancer for the treatment of NSCLC.
Signs and Symptoms
SCLC is usually diagnosed in patients in their sixth to seventh decade of life. Smoking is strongly associated with risk of developing lung cancer and particularly with SCLC. The relative incidence of SCLC has decreased over the last 4 decades reflecting decreases in smoking prevalence, changes to cigarettes, and reduced occupational hazards. There is a higher incidence of SCLC in males. However, the incidence gap between males and females has narrowed over the past 3 decades. Patients may have symptoms, signs, and/or laboratory test abnormalities caused directly by the primary tumor, those related to spread of tumor within the chest cavity or to distant sites like other organs, and those related to paraneoplastic syndromes.2-3 Paraneoplastic syndromes are group of rare disorders that are triggered by an abnormal immune system response to a cancerous tumor.4 Patients may present with symptoms of shortness of breath, cough, or chest pain due to disease in the chest. For patients with disease outside the chest, symptoms depend on the location of spread such as bone pain with bone metastases or headaches and dizziness in patients with central nervous system (CNS) disease involving the brain. Constitutional symptoms may include weakness, loss of appetite, weight loss or fever.3-5 These parameters alone do not help to clearly distinguish SCLC from NSCLC.
Before Treatment
Biopsy
 Before starting treatment, it is important to have a biopsy to confirm the diagnosis of SCLC. A biopsy is when a piece of tissue is taken and examined under a microscope. The type of biopsy performed depends on the location of the tumor. Most of the time, the patient does not require an overnight stay in the hospital to get a safe biopsy performed.
Before starting treatment, it is important to have a biopsy to confirm the diagnosis of SCLC. A biopsy is when a piece of tissue is taken and examined under a microscope. The type of biopsy performed depends on the location of the tumor. Most of the time, the patient does not require an overnight stay in the hospital to get a safe biopsy performed.
Many different methods may be used to obtain a biopsy, including:
A bronchoscopy: a patient is put to sleep with anesthesia and a very thin tube is inserted through the mouth or nose and gently slid into the main airways of the lungs. This procedure may be used to sample the lung tissue and/or a lymph node within the chest. An ultrasound may be used in an endobronchial ultrasound (EBUS) to assist in the procedure.
CT or ultrasound-guided biopsy: a needle guided by a CT scan or ultrasound is passed through the skin to obtain a piece of tissue.
A thoracentesis: in some patients presenting with fluid in the chest, a needle may be placed into the chest, and fluid is removed. The fluid is sent for evaluation of cancer cells.
Surgery: rarely, it is necessary to operate to obtain tissue to make a cancer diagnosis.
Although it is not common, if there is not enough tissue in the original biopsy, a second biopsy may be required.
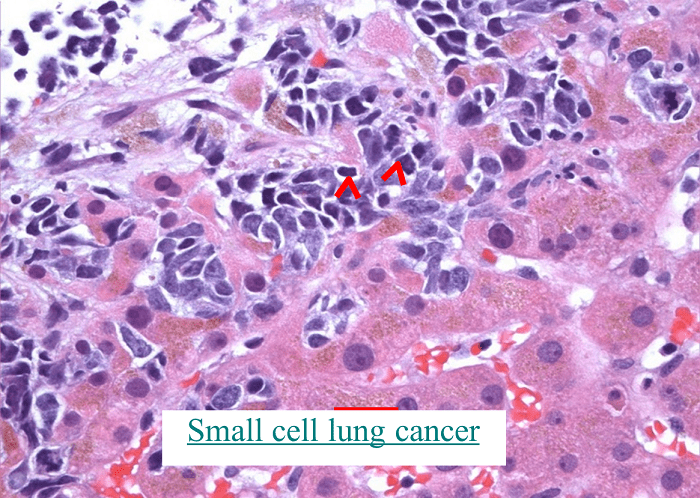
The biopsy specimen will be sent to a pathologist, a physician who specializes in looking at tissue samples. The pathologist will make the diagnosis and will determine what kind of cancer it is. SCLC may occur alone or combined with other tumors (Figure 1).
Figure 1. Microscopic image of a small cell lung cancer taken with a microscope
(The cancer cells are the small purple cells in the top half of the picture)
Radiology Tests
Once a diagnosis is established, radiograph imaging studies will be performed to determine the location of cancer within the body. The purpose of the testing is to stage the cancer, and define the extent of spread and establish the goal of treatment; if treatment will be curative or if a cure is not possible. Some of these tests may have been done prior to the biopsy and may not need to be repeated.
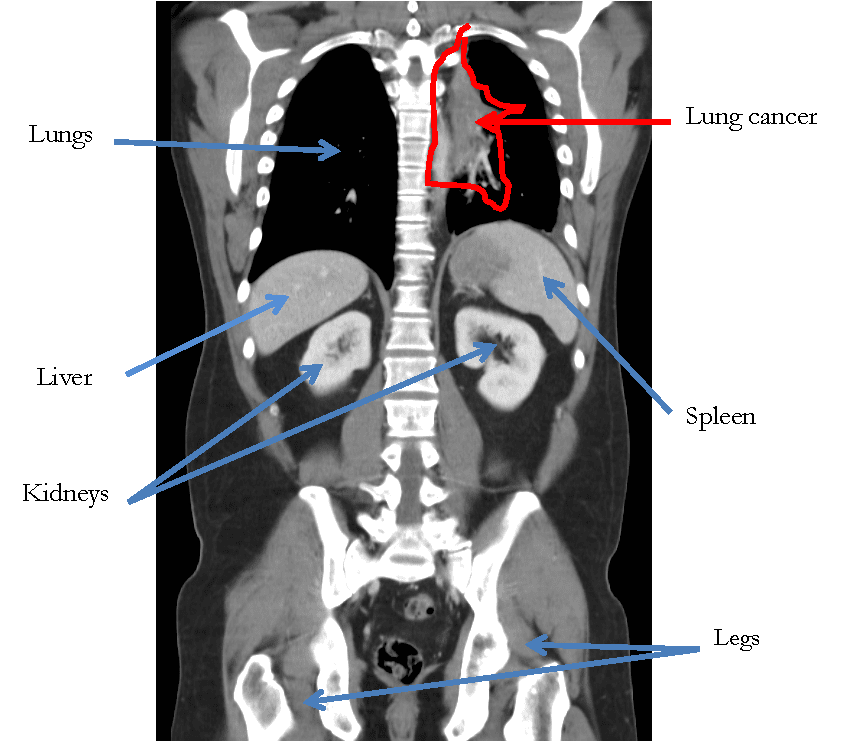
CT (CAT) scan of the chest, abdomen, and pelvis: this test has the purpose of looking for cancer that may be in the lungs, lymph nodes, liver, adrenal glands, bones, and other organs (Figure 2).
Figure 2. CT scan of the chest and abdomen with a cancer in the left lung.
Magnetic resonance imaging (MRI) of the brain: this test is done because SCLC may spread to the brain. Some patients cannot have an MRI scan and a CT scan with intravenous (IV) contrast of the brain may be done instead.
Bone scan: this test is done because SCLC may spread to the bones. If a patient is having a PET scan a bone scan is generally not necessary.
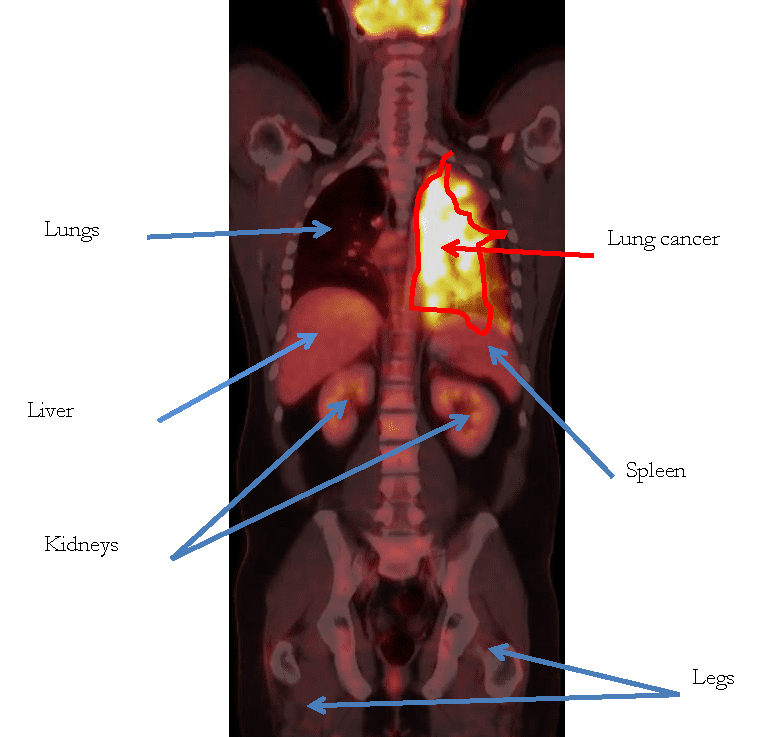
Positron Emission Tomography (PET) scan: because SCLC may spread to other parts of the body beyond the lung, this test is done to look at the entire body, except for the brain. PET scans measure increased levels of sugar (or glucose) that is being metabolized by the tumor as well some normal organs that may not have any active cancer (including the brain, heart, liver, and kidneys) (Figure 3).
Figure 3. Positron Emission Tomography (PET) scan which shows involvement of cancer in the left lung.
Blood Tests
Before starting treatment for lung cancer, blood tests are needed to evaluate how different organs in the body are working. The results may impact what treatment is prescribed. These tests are described below and are commonly performed in most patients with a cancer diagnosis and may be repeated regularly during or after treatment.
Kidney tests: creatinine and blood urea nitrogen (BUN).
Liver tests: alanine transaminase (ALT), aspartate aminotransferase (AST), alkaline phosphatase, and bilirubin.
Bone marrow tests: complete blood count.
Electrolytes: sodium, potassium chloride, phosphate and magnesium.
Staging
Stage is an indication of the size, location, and extent of the cancer. Approximately 60-70% of patients with SCLC are diagnosed with extensive stage (or stage IV) disease at presentation. This means that the tumor has spread outside of the chest. Although there is a shift in recommendations for the staging of SCLC, most patients are still diagnosed using the traditional staging system of limited stage disease and extensive stage disease.6 Patients with limited stage disease have cancer confined to the lung that can be treated with a combination of chemotherapy and radiation therapy and the goal of therapy is generally curative intent. In general, limited stage SCLC has a more favorable prognosis than extensive stage SCLC. Patients with extensive stage disease have cancer that has spread within or beyond the lung and the cancer and the goal of treatment is palliative (may not be cured with either systemic therapy or radiation therapy). Regardless of stage, patients with SCLC often experience an initial response to treatment.
The Tumor-Node-Metastasis (TNM) staging system is another system also used to stage SCLC. Using this system, SCLC may be staged as one of four stages, stage I, II, III which are similar to limited stage disease and stage IV which is the same as extensive stage disease. Staging is imperative to know prior to embarking on treatment. Staging is discussed in detail in Chapter 1: Diagnosing and Staging in Lung Cancer.
Treatment for Limited Stage Small Cell Lung Cancer
Surgery
Surgery is reserved for patients with small tumors confined to the lung and that have not spread to lymph nodes. Prior to surgery, lymph node sampling is performed via one of two methods:
Endobronchial ultrasound biopsy (EBUS) as mentioned above is a procedure where a tube is inserted through the mouth or nose into the airway and an ultrasound device is used to help identify lymph nodes that will be biopsied. This has become the preferred method to sample lymph nodes in patients with suspected cancers.
Mediastinoscopy is used in cases where lymph nodes cannot be accessed via EBUS. This is a minor surgery where a small incision is made at the bottom of the neck and a camera is placed into the chest to identify lymph nodes to biopsy.
If surgery is performed to remove the tumor from the lung, chemotherapy is recommended once the patient has recovered from surgery to treat microscopic disease, which are residual cancer cells that may be present but are not able to be seen with any imaging or the naked eye. If lymph nodes are found to be involved at the time of surgery, it will be necessary to have both chemotherapy and radiation after surgery. This situation is discussed further under the section on radiation therapy.
Due to the extensive and infiltrative nature of SCLC, surgery has not traditionally been a part of SCLC management. In less than 5% of cases, SCLC is diagnosed in early stages (stage I-IIA; T1-2, N0, M0) and surgery may be considered for those who have undergone sampling of the mediastinal (mid-chest) lymph nodes to prove that these are not involved.7 More commonly, limited stage SCLCs are stage IIB or beyond (T1-2, N1, M0). After completing investigations demonstrating no distant metastases (i.e., spread to the other lung or to other organs), these cases are typically treated with both chemotherapy and chest radiation given concurrently.

Combination of Chemotherapy and Radiation Therapy
Combined chemotherapy and radiation therapy is recommended for most patients with limited stage SCLC. Treatment is multidisciplinary. While a medical oncologist will oversee the chemotherapy, a radiation oncologist will oversee the radiation therapy during treatment.
If the patient is a current smoker, smoking cessation is very important prior to starting therapy. Several studies have suggested that patients who stop smoking prior to initiation of therapy have higher chances of cure and living longer compared to patients who continue to smoke.8 Patients can ask their doctors for resources to help them quit smoking. Additional information is available by calling 1-800-QUIT-NOW, reviewing https://smokefree.gov/, or Chapter 13: How to Quit Smoking Confidently and Successfully.
Chemotherapy
Chemotherapy is a type of drug that is used to kill cancer cells. Chemotherapy is an important component of treatment for patients with SCLC, whether given after surgery (i.e., adjuvant therapy), concurrently with radiation, or in extensive stage disease (discussed below).7
The most common chemotherapy regimen for the treatment of SCLC in the United States is a combination of a platinum agent , either cisplatin or carboplatin, plus etoposide.9 Your medical oncologist will choose between cisplatin or carboplatin by considering your other medical conditions and the different side effect profiles of each drug. Chemotherapy is given daily for 3 days in a row and repeated 3 weeks later. In between each cycle, the body recovers from the side effects of chemotherapy and gets ready for the next treatment. Each 3-week period is called a “cycle” and most commonly, a total of 4 cycles are given. Altogether, this means that chemotherapy treatment is given over a total of 12 weeks (3 weeks per cycle x 4 cycles). Concurrent chemotherapy and radiation for limited stage SCLC does not always require both to be started on exactly the same day. To prevent delays to treatment start, patients may receive 1-2 cycles of chemotherapy alone before radiation is added.
Chemotherapy is given intravenously through a needle that is inserted into the veins. Some patients may have an IV inserted into the vein that can be removed each day at the end of treatment while other patients may require a peripherally inserted catheter (PICC) or an implanted port that are placed in the vein and remains in the vein throughout the entire treatment.
The platinum and etoposide are both given on the first day and may take anywhere from 4-6 hours because additional fluids and medications are given prior to chemotherapy. Additional fluids are given prior to cisplatin chemotherapy due to the tendency of these agents to damage the kidneys without proper pre-hydration with fluids. These drugs can also cause nausea and vomiting, but pre-medications given before each treatment help to prevent these symptoms. Etoposide is given on its own on the second and third days, and takes about two hours to administer.
Most people do not feel anything unusual while receiving chemotherapy. However, patients may start to notice side effects 2-4 days after chemotherapy is administered.
Chemotherapy Side Effects
 Most patients do not have significant side effects from chemotherapy because of the many supportive therapies available. However, important side effects to remember include:
Most patients do not have significant side effects from chemotherapy because of the many supportive therapies available. However, important side effects to remember include:
Increased risk of infection: this is one of the most serious side effects of chemotherapy. Any patient with a fever (temperature higher than 100.4ºF), during chemotherapy needs to seek immediate medical attention.
Some patients may not have a fever and instead develop flu-like symptoms, cough, shortness of breath, pain with urination, diarrhea, or ear pain among other symptoms. You should seek medical attention if you have these symptoms as you may have an infection.
The increased risk of infection is because chemotherapy kills white blood cells, which normally are responsible for defending the body against infections. However, white blood cells grow back between each cycle. As a matter of precaution, it is important for patients to wash their hands regularly, avoid large crowds and sick people, and eat foods that come from trusted sources.
Fatigue: Both chemotherapy and radiation therapy may cause fatigue. This is normally the worst the first few days after chemotherapy and usually improves during the second and third week of each cycle. Fatigue can accumulate with each cycle of chemotherapy and radiation therapy. Fatigue will typically reach its worse level 1-2 weeks after the last dose of radiation treatment and then improve once all therapy is completed.
Nausea and Vomiting: As described above, nausea and vomiting have become manageable with the modern day use of potent anti-nausea medications. Medications are typically given in a preventative fashion and as needed. Pre-medications are given routinely before chemotherapy is administered regardless of whether patients have nausea/vomiting in order to prevent or reduce the risk of the development of any nausea/vomiting. In addition, patients are given medications to take home and use as needed should they require. These may include aprepitant or fosaprepitant which are given with chemotherapy as well as ondansetron, granisetron, dexamethasone, promethazine, prochlorperazine, metoclopramide and/or lorazepam which may be given with therapy or to take at home. Steroids are commonly included in the regimen your provider selects to prevent or reduce the risk of nausea and vomiting.
Hair Loss: Hair loss typically occurs within 3-4 weeks of starting chemotherapy. Hair often starts to grow back about one month after completing chemotherapy, but may be delayed in patients who are treated with radiation to the brain. The hair is often thicker and curlier than prior to treatment.
Other Side Effects: Other side effects of therapy include mouth sores, loss of appetite, diarrhea, easy bruising and bleeding (due to decreased hemoglobin and/or platelets), dehydration and damage to the kidneys, ringing in the ears (i.e., tinnitus) or numbness and tingling in the fingers and toes (i.e., peripheral neuropathy). All chemotherapy side effects should be discussed with a medical oncologist. See Chapter 8: Supportive Care.
Radiation Therapy
The techniques of radiation therapy for the treatment of limited stage SCLC are similar to what is done for stage IIIA to IIIB NSCLC—radiation therapy given concurrently with 4 to 6 cycles of chemotherapy. The same team approach is used in consultation, simulation, treatment planning, patient education, treatment delivery, and quality assurance. See Chapter 5: Radiation Therapy for Non-Small Cell Lung Cancer for additional technical details.
Unlike NSCLC, limited stage SCLCs are best treated with radiation twice a day using a lower dose per treatment, at least 6 hours apart, based on a famous clinical trial that showed patients with limited stage SCLC lived longer with twice a day treatments (4500 cGy in 150 cGy fractions given twice a day over 30 treatments in 3 weeks) compared to those getting treated once a day to the same total dose of 4500 cGy (given in 180 cGy fractions over 25 treatments). It should be noted that 4500 cGy given once daily over 25 treatments is considered suboptimal treatment and is never used in standard practice.10 However, treating a patient twice a day is harder on the patient due to increasing the daily treatment time (often burdening patients with more travel to and from the treatment center) and increasing the acute side effects. Therefore in practice, many radiation oncologists will treat patients to higher total doses of 6000 to 7000 cGy in 180 to 200 cGy fractions.11 A recent clinical trial showed that median survival was about the same for patients given 7000 cGy (30.5 months) given over seven weeks (35 once daily treatments) compared to twice daily treatment to 4500 cGy (28.7 months), supporting the use of higher dose once daily radiation treatments for patients with limited stage SCLC.12
Since 2004, the proportion of patients with SCLC getting once daily radiation treatments for SCLC treatment has been increasing and is now the preferred schedule compared to twice a day treatment.13 This schedule is less burdensome and less physically demanding for patients with limited resources.
As mentioned in the section on surgery, due to the extensive nature of SCLC and the fact that spread to the lymph nodes in the middle of the chest is so common, SBRT (stereotactic body radiation therapy as discussed in Chapter 5: Radiation Therapy for Non-Small Cell Lung Cancer) is not commonly offered, but is an acceptable and reasonable option for stage I-IIA patients who are either too frail for surgery or who refuse surgery.14Chemotherapy is recommended after SBRT.
Radiation Therapy After Surgery
When a patient is diagnosed with SCLC early enough to be stage I-IIA, they can be treated with surgery. As mentioned above in the section on surgery, if surgery is complete without involvement of lymph nodes, patients will be treated with chemotherapy afterwards. Nevertheless, situations where postoperative radiation is recommended after surgery include positive margins (the presence of cancer cells at the edge of where the surgeon excised the tumor) or any presence of cancer in the lymph nodes of the mediastinum (space in the mid chest containing the heart, esophagus, major blood vessels, and windpipe, N2 stage in the TNM staging system). Typically, a dose of 4500 to 5000 cGy is given in 180 cGy once-daily fractions over about 5 weeks. Chemotherapy can be given concurrently with radiation or sequentially. Radiation therapy may or may not be recommended if only the N1 lymph nodes (hilar, interlobar, lobar, segmental, or subsegmental—away from the mediastinum) are involved and depends on the discretion and expertise of the treatment team.15
Radiation Side Effects
Side effects of chest radiation in SCLC is similar to chest radiation in NSCLC.
Acute side effects occur when a patient is receiving lung radiation therapy with or without chemotherapy. These include redness and irritation of the skin overlying the radiation treatment portals; inflammation of the esophagus (esophagitis) causing heartburn or a feeling that something is stuck in the throat; irritation of the lung causing a dry cough; inflammation of the sac surrounding the heart causing chest pain (pericarditis); electric shock sensations in the low back or legs when bending the neck (Lhermitte sign); and generalized fatigue.
Fatigue can build week-to-week during a course of radiation therapy and may be at its worst up to 2 weeks following the last radiation therapy. This sensation of fatigue is due to treatment not necessarily from the tumor itself.
These acute side effects typically begin to resolve 2 weeks after completing chest radiation therapy.
Subacute side effects occur 1 to 6 months after completing radiation therapy. These side effects are less frequent and may include radiation pneumonitis, which is inflammation of the lung that causes chest pain, fever, and a dry cough.16 As mentioned above in the section on treatment planning, radiation pneumonitis occurs infrequently, especially when the V20 (volume of both lungs receiving ≥ 20 Gy or 2000 cGy) is no more than 35%. Your radiation oncologist, dosimetrist, and physicist work hard to ensure that the least amount of radiation possible goes to normal lung without sacrificing coverage of the lung tumor. Treatment of radiation pneumonitis includes corticosteroids such as prednisone or dexamethasone.
Long term side effects of lung radiation therapy include pulmonary fibrosis (permanent scarring of the radiated lung tissue), esophageal fibrosis and stricture (scarring and narrowing of the esophagus that causes difficulty swallowing and treated with esophageal dilation), constrictive pericarditis (shrinkage of the sac surrounding the heart, that may require surgical removal), and damage to the heart muscle and blood vessels that may increase the risk of heart failure and heart attack. These long term side effects are uncommon because modern radiation therapy techniques have resulted in better sparing of normal tissues and organs. Excerpted from Chapter 5: Radiation Therapy for Non-Small Cell Lung Cancer.
Prophylactic Cranial Irradiation
Because the risk of brain metastases in SCLC is significant, MRI of the brain with contrast is part of the staging workup for limited stage small cell lung cancer. About 20% of SCLC patients will already have brain metastases when they are diagnosed.17 Prophylactic cranial irradiation (PCI) is giving radiation therapy to the entire brain (also called whole brain radiation therapy, WBRT) for patients without brain metastases on MRI to lower the risk of developing brain metastases (59% get brain metastases without PCI while 33% get brain metastasis with PCI over 3 years in patients with limited stage SCLC).18 The standard dose has been 2500 cGy given in 250 cGy fractions over 10 treatments. PCI includes the entire brain (like whole brain radiation) but also includes the spinal cord down to the level of the C2 vertebra. PCI is currently recommended for all patients with limited stage SCLC who have a good response to chemotherapy and radiation.
If you chose to receive PCI, then it is important to be aware that one of the major side-effects of this treatment is a dementia-like syndrome. One recent therapy to perhaps prevent or decrease this syndrome is for your radiation oncologist to use a technique called hippocampal sparing. The hippocampus are 2 very small components of our brain located on the right and left side. If these small anatomical areas can be blocked during radiation, the dementia syndrome may be lessened. In addition, there is a pill called Memantine which can be given over a 6-month time frame which may also prevent the dementia-like syndrome for occurring in some patients.
Nevertheless, your radiation oncologist should thoroughly discuss the benefits and risks of whole brain radiation with you before deciding on this (see the section on palliative radiation to the brain in Chapter 5: Radiation Therapy for Non-Small Cell Lung Cancer).
If you choose not to receive PCI, then an MRI of the brain should be done every 3 to 4 months to monitor you closely.
Follow Up
After therapy is completed, patients are followed regularly to make sure they recover from side effects and also for surveillance to monitor for signs of disease recurrence.
The majority of side effects usually start to improve about four weeks after completing chemotherapy with more energy and increased appetite. However, prophylactic cranial irradiation, which starts about a month after chemotherapy is complete, may delay recovery. Late side effects from radiation to the chest may occur such as inflammation of the lungs (pneumonitis) or difficulty swallowing from narrowing of the esophagus, both of which can be effectively treated. For this reason, it is important to tell the doctors about any new symptoms that occur after treatment has ended.
Regular visits are recommended with the medical oncologist every one to two months initially, and then less frequently if the patient is feeling well. Your radiation oncologist will want to see you one to three months after completing your treatment and subsequently at longer intervals in coordination with your medical oncologist. At every visit the doctor will review symptoms and perform a physical examination. A CT scan of the chest and blood work may be done every three to four months initially, every four to six months later, and annually after three years. An MRI (preferred) or CT of the brain will be done every three to four months during the first year, then every six months. Regular follow-up and routine CT/MRI scans are part of surveillance, which is scheduled monitoring for early signs of disease recurrence.
Prognosis
The intention of treatment for limited stage SCLC is to cure the cancer. It is expected that 70-90% of patients who receive therapy will have a response with the cancer showing shrinkage. However even after completing treatment, there is a high risk of recurrence and only 25-30% of patients are alive at five years. For patients who do not wish to receive treatment or are unable to receive treatment, the expected survival is lower.
Treatment for Extensive Stage Small Cell Lung Cancer
Patients with extensive stage SCLC have cancer that has spread either within or beyond the lungs. For the first time in three decades we have seen progress in the treatment of patients with extensive stage disease with a combination of chemotherapy and immunotherapy. Radiation therapy may be used for consolidation or for symptom control during or after chemotherapy. Surgery is generally not recommended.
Chemotherapy
Similar to patients with limited stage disease, the chemotherapy backbone consists of a platinum doublet cisplatin or carboplatin with etoposide in combination with immunotherapy for patients in North America and Europe. Treatment is given over 3 days with platinum and etoposide given on the first day along with atezolizumab or durvalumab and then etoposide alone on day 2 and 3 for four cycles, with each treatment being 21 days apart. After four cycles atezolizumab or durvalumab is continued as a maintenance therapy for as long as the treatment remains effective. Atezolizumab and durvalumab are drugs known as a checkpoint inhibitors (see Immunotherapy below).
Treatment is given through a vein using either a needle inserted into the arm or a PICC or PORT as described above. Treatment is generally given as an outpatient and may take 4-6 hours on the first day and then about two hours on day two and three. Prior to treatment on the first day blood work is done to make sure it is safe to give chemotherapy and you will likely meet with your medical oncologist or a member of the healthcare team. In between each cycle the body recovers from the side effects of chemotherapy and immunotherapy and gets ready for the next treatment. Chemotherapy for extensive stage disease is usually given for 4 cycles with immunotherapy, followed by maintenance immunotherapy every 3 weeks with imaging performed every 2-3 cycles.
 Immunotherapy
Immunotherapy
Immune checkpoint inhibitors have been approved for the treatment of multiple different tumor types including patients with SCLC. These agents work very differently from chemotherapy acting on proteins expressed on cancer cells and T cells that normally prevent the body from recognizing cancer cells as foreign. When these proteins are blocked the T cells are activated and able to kill cancer cells. Currently, atezolizumab and durvalumab are approved in combination with chemotherapy as a first-line option for patients with SCLC.19-20 Not all patients are candidates for treatment with immunotherapy. These agents are generally not recommended for patients with a history of uncontrolled autoimmune disease, organ transplant or certain paraneoplastic syndromes.19-20
Chest Radiation
The combination of chemotherapy and radiation therapy is not typically given to patients with extensive stage SCLC. However, radiation may be given to help relieve symptoms, such as pain, shortness of breath, difficulty swallowing, and/or blockage of a bronchus, while a patient waits to get started with chemotherapy. SCLC is the most common cause (75 to 80%) of superior vena cava (SVC) syndrome, a condition caused by a large tumor compressing the superior vena cava causing swelling of the face, neck, and upper arms; shortness of breath; and coughing.23 Chest radiation in extensive stage SCLC is given on a case by case basis, and when given, can sometimes be given after a few courses of chemotherapy to treat any tumor that is left. Despite the fact that chest radiation is not used routinely in extensive stage SCLC, clinical studies analyzing the benefits of chest radiation in patients with extensive stage SCLC patients have been promising and have suggested that patients may live longer if chest radiation is added to chemotherapy.24
Prophylactic Cranial Irradiation (PCI)
MRI of the brain with contrast is also part of the staging workup for extensive stage SCLC. Prophylactic cranial irradiation (PCI, 2500 cGy given in 250 cGy fractions over 10 treatments as in limited stage SCLC) was a standard treatment for patients with extensive stage SCLC patients without brain metastases on MRI to lower the risk of developing brain metastases (40.4% to 14.6% over 1 year in extensive stage), as mentioned in Lung Cancer Choices, 5th Edition.25 However, another clinical trial investigating PCI showed that there was no improvement in survival with PCI for patients with extensive stage SCLC who responded well to chemotherapy.26 Because of the side effects to the brain and the limited survival benefit, patients with extensive stage SCLC can choose to omit PCI. Whether or not PCI is given, you should get an MRI (preferred) or CT of the brain every three to four months to monitor for the appearance of any brain metastases. It must be emphasized that PCI is still recommended for patients with limited stage SCLC.
Management of Metastases with Radiation
Brain Metastases
When patients with SCLC develop brain metastases, whole brain radiation (WBRT) is the recommended radiation treatment method, for a total dose of 3000 cGy given in 300 cGy fractions over 10 treatments. If patients have a good performance status and have an estimated survival of 6 months or more, some patients can be treated with a longer fractionation dose of 3750 cGy in 250 cGy fractions over 15 treatments. Memantine, a drug FDA approved to treat Alzheimer’s dementia, has been shown to reduce the rate of decline in memory, executive function, and processing speed in patients receiving WBRT.27 Ask your radiation oncologist about a prescription for memantine if you are going to receive PCI or WBRT.
Even when a limited number of brain masses are detected on MRI, it is understood that in SCLC, there may be many additional tumors that are too small to be detected on MRI. Because of this, focused treatments such as surgical removal or stereotactic radiosurgery (SRS) have not been routinely offered in the past, as it is felt that the entire brain is at risk. Nevertheless, SRS for brain metastases in SCLC has been studied and shows promise as an alternative to whole brain radiation.28 Further studies evaluating SRS for brain metastases in SCLC are currently being conducted.29
In patients that develop brain metastases after PCI, repeat WBRT can be given safely but is associated with more cognitive side effects. As an alternative to repeat WBRT, SRS can also be done with potentially less side effects provided there are a limited number of metastases.30
Metastases to Other Organs
SCLC can also spread to the liver, adrenal glands, bones (including vertebra). In these cases, radiation therapy (either conventional radiation or SBRT) can be used to treat these sites to relieve pain and prevent fracture (in cases of bone metastases). SRS and SBRT are discussed in detail in Chapter 5: Radiation Therapy for Non-Small Cell Lung Cancer in the section “Palliative Radiation Therapy for Sites of Metastases in Stage IV Non-Small Cell Lung Cancer”.
Prognosis
Patients with extensive stage SCLC are generally not cured with chemotherapy, immunotherapy, or radiation therapy. The purpose of treatment is to improve quality of life and symptoms as well as prolong survival. Approximately 60-70% of patients will respond to chemotherapy the first time with their cancer shrinking or disappearing. Unfortunately, responses may not last for long and additional chemotherapy or radiation may be needed. For patients who get treated, with chemotherapy and immunotherapy, the 3-year survival is 17.6%.31 Life expectancy is usually less than 2-3 months and often only a few weeks for patients who choose not to have any treatment.
Clinical Trials
 Given the limited number of drugs currently approved for SCLC, not due to lack of studies but due to lack of success, clinical trials remain critical to advance therapy for this disease.
Given the limited number of drugs currently approved for SCLC, not due to lack of studies but due to lack of success, clinical trials remain critical to advance therapy for this disease.
A clinical trial is a research program designed to evaluate whether a new drug is effective in the treatment of a particular disease. Some trials may compare a new treatment to the current standard of care or add a treatment to the standard of care. Some people may not wish to participate in a clinical trial for fear of not receiving adequate treatment for their disease, however this is not the case in clinical trials involving patients with cancer.
Patients should speak with their doctor about a clinical trial, ask questions, and decide if the trial is a reasonable choice for their disease. Sometimes a patient may not be able to participate in a trial because of other comorbid illnesses and the doctor would know if this was the case.
Patient participation in a clinical trial is critical to advancing the care for patients with lung cancer. See Chapter 7: Clinical Trials and Emerging Therapies for Lung Cancer

Questions to Ask Your Treatment Team
References
- American Cancer Society, Facts & Figures: 2023 https://acsjournals.onlinelibrary.wiley.com/doi/10.3322/caac.21763 [Last accessed 12/1/2023]
- Beckles MA, Spiro SG, Colice GL, Rudd RM. The physiologic evaluation of patients with lung cancer being considered for resectional surgery. 2003;123(1 Suppl):105S-114S.
- Jackman DM, Johnson BE. Small-cell lung cancer. Lancet. 2005;366(9494):1385-1396.
- Yang P, Allen MS, Aubry MC, et al. Clinical features of 5,628 primary lung cancer patients: experience at Mayo Clinic from 1997 to 2003. 2005;128(1):452-462.
- Chute CG, Greenberg ER, Baron J, Korson R, Baker J, Yates J. Presenting conditions of 1539 population-based lung cancer patients by cell type and stage in New Hampshire and Vermont. 1985;56(8):2107-2111.
- Byers LA, Rudin CM. Small cell lung cancer: where do we go from here? 2015;121(5):664-672.
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology. Small Cell Lung Cancer Version 2.2017. 2016 Sept 15;National Comprehensive Cancer Network. [Abstract available at https://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf.
- Videtic GM, Stitt LW, Dar AR, et al. Continued cigarette smoking by patients receiving concurrent chemoradiotherapy for limited-stage small-cell lung cancer is associated with decreased survival. J Clin Oncol. 2003;21(8):1544-9.
- Sundstrom S, Bremnes RM, Kaasa S, et al. Cisplatin and etoposide regimen is superior to cyclophosphamide, epirubicin, and vincristine regimen in small-cell lung cancer: results from a randomized phase III trial with 5 years’ follow-up. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 2002;20(24):4665-4672.
- Turrisi AT, 3rd, Kim K, Blum R, et al. Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med. 1999;340(4):265-271.
- Travis WD. Advances in neuroendocrine lung tumors. Annals of oncology: official journal of the European Society for Medical Oncology / ESMO. 2010;21 Suppl 7:vii65-71.
- Jeffrey A Bogart, Xiaofei F. Wang, Gregory A. Masters, et al. Phase 3 comparison of high-dose once-daily (QD) thoracic radiotherapy (TRT) with standard twice-daily (BID) TRT in limited stage small cell lung cancer (LSCLC): CALGB 30610 (Alliance)/RTOG 0538. Journal of Clinical Oncology 2021 39:15_suppl, 8505-8505.
- Kazemi M, Ladbury C, Liu J, Glaser S, Williams T, Amini A. Thoracic Radiation in Limited Stage Small Cell Lung Cancer: Trends in Radiation Fractionation. Clin Lung Cancer. 2023 Jun;24(4):322-328.
- Rathod S, Koul R, Bashir B, et al. Role of Stereotactic Body Radiation Therapy in Early Stage Small Cell Lung Cancer in the Era of Lung Cancer Screening: A Systematic Review. Am J Clin Oncol. 2019 Feb;42(2):123-130.
- NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Small Cell Lung Cancer V.3.2023. © National Comprehensive Cancer Network, Inc. 2022. All rights reserved. Accessed August 26, 2023. To view the most recent and complete version of the guideline, go online to NCCN.org.
- Auperin A, Arriagada R, Pignon JP, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. Prophylactic Cranial Irradiation Overview Collaborative Group. N Engl J Med. 1999;341(7):476-484.
- Seute, T., Leffers, P., ten Velde, G.P., Twijnstra, A. Detection of brain metastases from small cell lung cancer: consequences of changing imaging techniques (CT versus MRI). 2008;112:1827–1834
- Chang JY, Bradley JD, Govindan R, Komaki R. Toxicity of Normal Tissue. In: Halperin EC, Perez CA, Brady LW, eds. Principles and Practice of Radiation Oncology. Philadelphia, PA: Lippincott Williams & Wilkins; 2008:1102-4.
- Horn, L, Liu SV2, Atezolizumab plus Chemotherapy in Small-Cell Lung Cancer. N Engl J Med. 2019 Feb 28;380(9):889-890. doi: 10.1056/NEJMc1900123.
- Paz-Ares et al. Durvalumab plus platinum-etoposide versus platinum-etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): a randomised, controlled, open-label, phase 3 trial. Lancet. 2019, 394 (10212): 1929-1939.
- Ready N, Farago AF, de Braud F, Atmaca A, Hellmann MD, Schneider JG, et al. Third-Line Nivolumab Monotherapy in Recurrent SCLC: CheckMate 032. J Thorac Oncol 2019;14(2):237-44 doi 10.1016/j.jtho.2018.10.003
- Chung HC L-MJ, Kao SCH, Miller WH, Ros W, et al. Phase 2 study of pembrolizumab in advanced small cell lung cancer (SCLC): KEYNOTE-158. ASCO abstract 850620
- emedicine > Superior Vena Cava Syndrome. Author: Michael S Beeson, MD, MBA, FACEP, Professor of Emergency Medicine, Northeastern Ohio Universities College of Medicine and Pharmacy; Attending Faculty, Summa Health System. Updated: Dec 3, 2009.
- Palma DA, Warner A, Louie AV, Senan S, Slotman B, Rodrigues GB. Thoracic Radiotherapy for Extensive Stage Small-Cell Lung Cancer: A Meta-Analysis. Clinical Lung Cancer. 2016;17(4):239-244.
- Slotman B, Faivre-Finn C, Kramer G, et al. Prophylactic cranial irradiation in extensive small-cell lung cancer. N Engl J Med. 2007;357(7):664-672.
- Takahashi T, Yamanaka T, Seto T, Harada H, Nokihara H, Saka H, Nishio M, Kaneda H, Takayama K, Ishimoto O, et al. Prophylactic cranial irradiation versus observation in patients with extensive-disease small-cell lung cancer: a multicentre, randomised, open-label, phase 3 trial. The Lancet Oncology. 2017;18(5):663–71.
- Brown PD, Pugh S, Laack NN, et al. Memantine for the prevention of cognitive dysfunction in patients receiving whole-brain radiotherapy: a randomized, double-blind, placebo-controlled trial. Neuro Oncol. 2013 Oct;15(10):1429-37.
- Jiang W, Haque W, Verma V, Butler B, Teh BS. Stereotactic radiosurgery for brain metastases from newly diagnosed small cell lung cancer: practice patterns and outcomes. Acta Oncol. 2019 Apr;58(4):491-498.
- NRG Oncology. (2021). Phase III Trial of Stereotactic Radiosurgery (SRS) versus Hippocampal-Avoidant Whole Brain Radiotherapy (HA-WBRT) for 10 or Fewer Brain Metastases from Small Cell Lung Cancer. (NRG CC-009). Retrived from https://www.nrgoncology.org/Clinical-Trials/Protocol/nrg-cc009-1
- Nakazaki K, Higuchi Y, Nagano O, et al. Efficacy and limitations of salvage gamma knife radiosurgery for brain metastases of small-cell lung cancer after whole-brain radiotherapy. Acta Neurochir. 2013;155:107–113.
- Pas-Ares et al. Durvalumab, with or without tremelimumab, plus platinum-etoposide in first-line treatment of extensive-stage small-cell lung cancer: 3-year overall survival update from CASPIANESMO Open. 2022 Apr; 7(2): 100408.

