Lung Cancer Choices© 6th Edition Menu
Chapter 1: Diagnosis and Staging of Lung Cancer
Chapter 2: Comprehensive Biomarker Testing
Chapter 3: Surgery for Lung Cancer Patients
Chapter 5: Radiation Therapy for Non-Small Cell Lung Cancer
Chapter 6: Treatment for Small Cell Lung Cancer
Chapter 7: Clinical Trials and Emerging Therapies for Lung Cancer
Chapter 9: Nutrition in the Patient with Lung Cancer
Chapter 10: Sexuality and Lung Cancer
Chapter 11: Integrative Medicine, Complementary Therapies, and Chinese Medicine in Lung Cancer
Chapter 12: Lung Cancer in People who have Never Smoked
Chapter 13: How to Quit Smoking Confidently and Successfully
Lung Cancer Question Builder - Develop your list of "Questions to Ask" your providers
Nutrition in the Patient with Lung Cancer
Rhone M. Levin, M.Ed, R.D.N., C.S.O., L.D.
Introduction
Nutrition is vital in cancer treatment and survivorship. Food provides the building blocks needed by the body to repair and heal from cancer therapy. The benefits of proper nutrition during cancer treatment include improved quality of life, decreased discomfort from side effects, and reduced frequency of complications and treatment breaks.
Lung cancer treatment can create a burden of healing that can overwhelm even a healthy patient’s nutritional reserve. Cancer itself can affect appetite, digestion, and use of nutrients. Treatment regimens such as surgery, chemotherapy, immunotherapy, and radiation can cause side effects that interfere with eating and drinking. A patient’s nutritional status can deteriorate throughout treatment, reducing their ability to tolerate treatment. Decisions about treatment regimen may be determined based on general health performance status scores.1 Weight loss and decreased ability to consume adequate nutrition can negatively influence those performance scores and alter treatment options.2
Many people that begin lung cancer treatment have already experienced some decreased appetite (anorexia) and may have noticed an early sense of fullness when eating (early satiety). Anorexia is often described as a disinterest in food. The effects of anorexia and early satiety can lead to unintentional weight loss and possibly even malnourishment.
Among patients with advanced lung cancer, more than half will become malnourished during treatment. Malnourishment is associated with worse outcomes in patients treated for cancer because nutritional deficiencies can decrease treatment tolerance, response to therapy, quality of life, and even survival.3 Taking action to improve nutritional status may improve strength, energy level, and protect quality of life.
The goal of nutrition in treatment is to support the healing process. In this chapter, we review the factors that affect nutrition and healing; the common barriers to eating, effective strategies to manage the side effects that limit nutritional intake; and resources to use during treatment and survivorship.
Protecting Lean Body Mass
During recovery from cancer treatment, it is important to maintain muscle or “lean body mass” to help maintain optimal health and allow you to participate in usual activities. Muscle wasting can result in debilitation, decreased functional status, and reduced quality of life.4 Maintenance of lean body mass during treatment may even improve response to cancer treatment.5-6
For some people, the first sign of illness may be an unexpected or involuntary weight loss. Some people may have reacted to this weight loss and decreased interest in food with a happy exclamation, “Oh good – I’ve been trying to lose weight!” or “My doctor told me I should lose 20 pounds”. Patients may allow the weight loss to continue, believing that the process must eventually stop. However, weight loss with a diagnosis of lung cancer is different than intentional weight loss that occurs when dieting. Both underweight and overweight people are at risk for malnourishment during lung cancer therapy.5-6
Involuntary weight loss may occur in about half of the people with lung cancer, even a weight loss of 5% may influence health outcomes. Some involuntary muscle loss occurs when people feel ill and cannot eat enough to maintain their weight. As skeletal muscle is lost, patients experience fatigue, lack of energy for daily activities, decreased ability to move with balance and safety, and decreased ability to cough and clear pulmonary secretions. As smooth muscle is lost, a person may have delayed stomach emptying and feel satiated or feel full; there may be decreased digestion associated with increased nausea as well as a loss of cardiovascular function associated with lightheadedness or dizziness.7
 Cancer cachexia is a syndrome that results in a progressive loss of muscle mass and fat stores and leads to progressive functional impairment. It is associated with a lack of appetite and negative energy and protein balance. It is important to identify these symptoms, called “pre-cachexia”, and take action to treat any side effects or barriers to eating.8 Medications, such as appetite stimulants, may help manage cancer cachexia, and this option may be discussed with the physician.9
Cancer cachexia is a syndrome that results in a progressive loss of muscle mass and fat stores and leads to progressive functional impairment. It is associated with a lack of appetite and negative energy and protein balance. It is important to identify these symptoms, called “pre-cachexia”, and take action to treat any side effects or barriers to eating.8 Medications, such as appetite stimulants, may help manage cancer cachexia, and this option may be discussed with the physician.9
Nutritional counseling which focuses on food choice and behaviors related to eating is effective in addressing lung cancer malnutrition. Medical Nutrition Therapy, a technique used by a Registered Dietitian/Nutritionist (RDN), can help patients to increase protein and calorie intake, improve weight status, enhance energy and strength, and protect quality of life in people undergoing cancer treatment.7, 10-12 Many cancer centers have specially trained RDNs who are dedicated to the nutritional care of cancer patients. If the cancer center does not have an Oncology RDN, a referral may be obtained from the doctor. Contact the Commission on Dietetic Registration to find a board-certified specialist in oncology nutrition – who has the credential “CSO”.
Nutrition and Healing
Each time a patient receives treatment for cancer – surgery, chemotherapy, targeted therapy, immunotherapy, or radiation therapy – the body responds to the treatment with healing. The healing process requires specific nutrients, extra calories, and additional protein. People receiving treatment for lung cancer may require more energy and protein than when they were not sick; this state of increased demand for calories and protein is termed “hyper-metabolic.”13-15
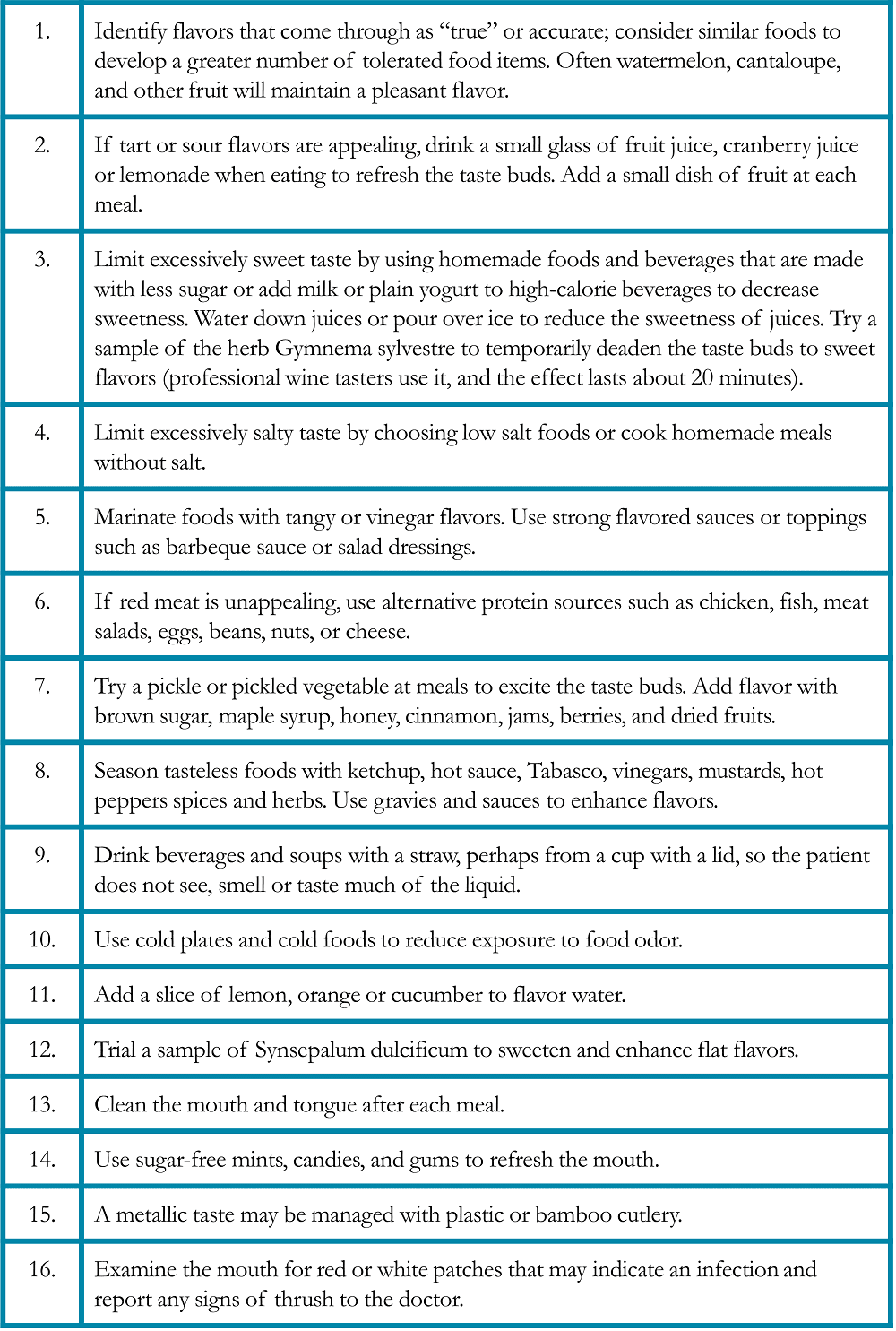
Total daily caloric content should be considered in addition to total daily protein intake. If weight loss continues despite higher protein intake, the protein will be utilized for calories and will not be available for structural repair. It is useful to have a general expectation for the number of calories and protein required each day (Table 1). Your individual needs may differ, ask your healthcare team about your specific needs.
Table 1. Caloric and Protein Requirements during healing in Patients Treated for Lung Cancer*
Hydration and Fluid Balance
Staying hydrated is important to feeling well during treatment. Hydration balance is cumulative, and it can take several days to become dehydrated or to re-acquire adequate hydration status.
Fluid needs may be increased due to chemotherapy, fever, perspiration, diarrhea, use of oxygen, or the presence of chronic obstructive pulmonary disease (COPD). Symptoms of dehydration include: fatigue, thirst, dry mouth, decreased urine output, concentrated or darker colored urine, decreased skin turgor (elasticity of the skin), headache, and dizziness. Mild chronic dehydration may also increase fatigue and contribute to constipation. A fluid deficit of 1% body weight may decrease metabolic function by 5%.16
Patients may consider tracking daily fluid intake to ensure adequate hydration. It may help to measure favorite cups and mugs to make it easier to estimate the volume of fluid consumed. It is best to drink fluids throughout the day, drinking half of their fluid requirements during the first half of the day. Some patients prefer to plan their fluid intake by the hour, such as: drink 1 cup per hour, throughout the day. Most liquids may be included as part of daily hydration, including flavored waters, milk, juice, smoothies, milkshakes, and soda. Caffeinated beverages may be included as part of daily fluid intake if caffeine consumption is moderate (less than 300 mg per day which is the equivalent of 2 cups of coffee). Caffeine may cause the stomach to empty faster and therefore, may be dehydrating.
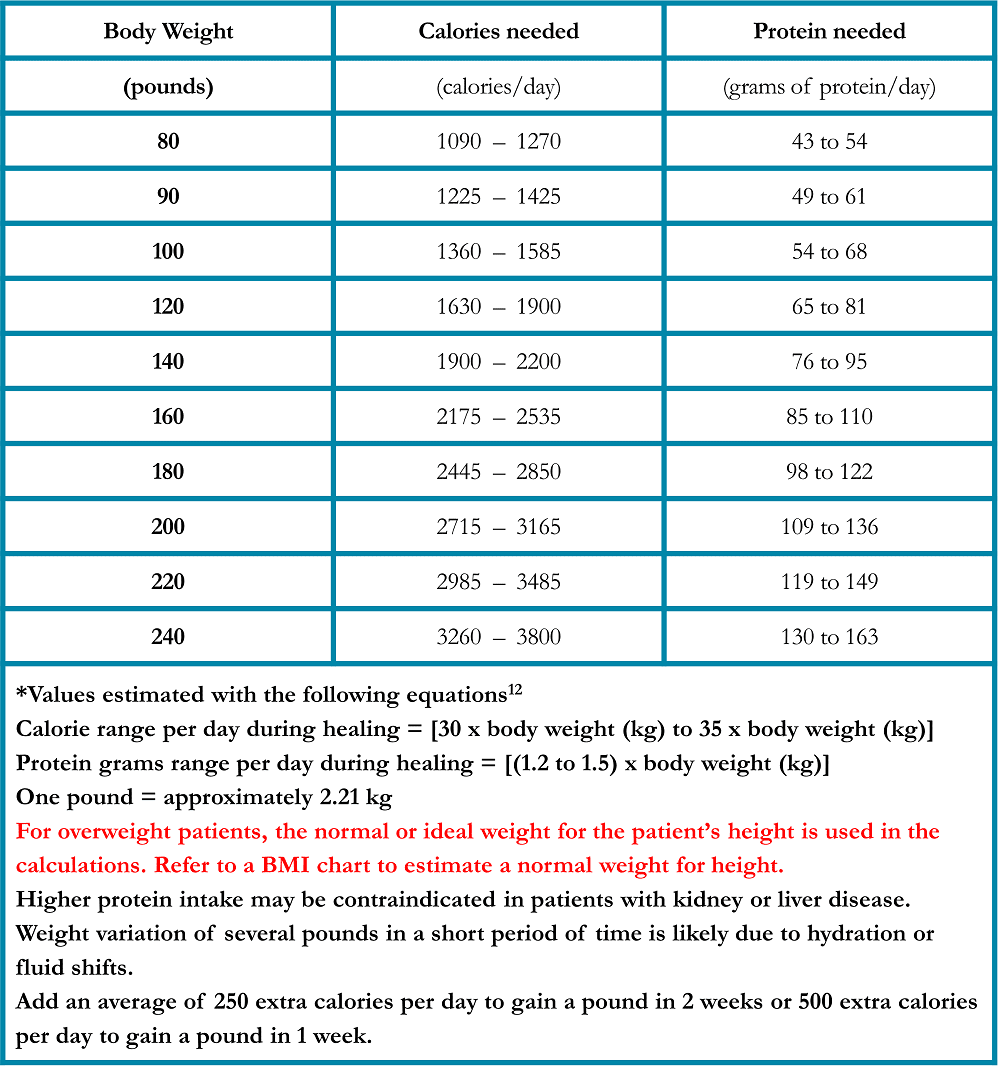
Many foods such as fruits, soups, gelatin, ice cream, and frozen desserts include hydrating fluids. Fluids intended for rehydration, called “sports drinks,” have a small amount of carbohydrates and electrolytes to help them absorb more effectively. Oral rehydration salt solutions (or “ORS” products) are effective at improving hydration status and are now readily available in pharmacies. Ask your healthcare team if ORS would be appropriate for you. Daily fluid requirements may be estimated using the chart below (Table 2). Add 1 cup for each episode of diarrhea. Your individual needs may differ, ask your doctor about your specific needs.
Table 2. Fluid Requirements During Healing in Patients Treated for Lung Cancer*
Strategies to Help Lung Cancer Patients Eat Enough Food
The nutrition focus during lung cancer treatment is to get enough calories and protein to support the healing process. Oncologists recommend that “all calories are good calories in lung cancer therapy.” The aim is to make eating as tolerable and interesting as possible and remove any unnecessary diet restrictions. Information found on the television, in magazines, and on the internet regarding “good nutrition” may not apply to people going through cancer treatment. Ask your healthcare team’s advice if you have been following a heart-healthy, low-fat, low-cholesterol, or low-carbohydrate diet.
Be Flexible
Cultural traditions regarding “what makes a meal” may need to be modified in treatment, such as changing expectation of eating three large meals a day, to planning six small meals instead. If simple foods are tolerated better, the patient may consider using non-traditional meal choices; such as pancakes for lunch and scrambled eggs at the evening meal. Snacking has been found to increase total intake without affecting meal intake, especially if snacks are timed approximately two hours before the next meal.17
A quality snack may be created by combining any two of the following food groups: breads/starches; meats/nuts/beans/eggs; milk/cheese/yogurt dairy products; fruits/vegetables (Table 3).
Table 3. Examples of Good Quality Snacks

This technique provides a combination of carbohydrates, proteins, and fats. The goal for a good quality snack (or small meal) is about 250 calories and about 6 grams of protein. Some patients prefer to drink their calories when their appetite is poor. Beverages that contain calories and protein can be used as a snack by itself, or as a meal replacement.
Making Every Bite Count
Many foods and beverages are available in full fat or high-calorie options (for example choosing whole milk instead of skim milk). Some can be enhanced to increase nutrient density by adding protein powder supplements or calorie enhancers (for example, adding cream to a milkshake instead of milk). Using more fat in dishes may be helpful for those who experience dyspnea (shortness of breath). Fat requires less oxygen in the digestion process; thus, higher-fat meals may minimize oxygen requirements.
As appetite may decrease with cancer treatment, using more fat is an effective way to maximize caloric intake. Some people who have followed a low cholesterol diet must rediscover fat-containing foods. Monounsaturated fats or “heart-healthy” fats may be emphasized, such as olive, avocado, nut, or oily fish, to achieve a higher caloric density. Some physicians will allow all fat-containing foods during cancer treatment to aid in the taste and palatability of dishes.
Each teaspoon of any fat, oil or butter contains about 45 to 50 calories. By adding one teaspoon of fat to each meal and snack, caloric intake is increased by approximately 250 calories per day without having to eat a larger volume of food. Another strategy is to add one to two tablespoons of heavy cream to any milk-containing food or beverage, thus increasing the calorie content of that food by approximately 50-100 calories. These additions are almost invisible to the person who is trying to maximize caloric intake.
Diabetic Concerns
Many people are required to follow a diabetic diet that limits carbohydrate intake. When appetite is decreased, and meal size is reduced, diabetic diets may be liberalized to allow the addition of carbohydrates. Carbohydrate counting or substitution may help increase caloric intake. Eating carbohydrates may be an uncomfortable idea for patients who have followed their doctor’s advice for many years to avoid simple sugars and starches. Many doctors also liberalize the blood glucose goals of patients during cancer treatment and may consider using medication to manage blood glucose—not food restriction.
A common strategy for people with diabetes to maximize their oral intake is to have both low carbohydrate and regular carbohydrate foods available. If eating is minimal, the food item with full carbohydrate content may be used. If consumption is close to usual portion sizes and frequency, the lower carbohydrate version may be used. An example of food substitution can be made regarding yogurt: choose a full carbohydrate version when it is the only food eaten for lunch but choose a low sugar yogurt if it is consumed with a sandwich and bowl of soup.
Be aware of the symptoms of low blood glucose in patients who take some diabetic medications, as decreased oral intake while continuing to take insulin may cause low blood sugar or hypoglycemic episodes. These symptoms may include lack of concentration, clammy sweats, shaking or tremors, changes in vision, lightheadedness, or dizziness. If any of these symptoms occur, blood glucose level should be tested immediately, and if low, carbohydrates should be provided. Strategies to prevent hypoglycemic episodes include eating and drinking small amounts more frequently during the day; planning an evening snack before going to sleep; and discussing modifications of medication with your healthcare team. Diabetic patients may also consider carrying glucose tablets or hard candy and keeping some fruit juice at home to drink if blood sugar drops.
Vitamins and Mineral Supplements
Several studies have examined the use of supplemental antioxidants, vitamins, and minerals in patients with advanced non-small cell lung cancer receiving chemotherapy. Most studies have not shown the protective benefit of antioxidants during treatment, nor reduction in cytotoxic side effects.18 The VITAL Study (Vitamins and Lifestyle Study) determined that people at risk for developing lung cancer, particularly smokers, should not use beta carotene supplements, retinol or lutein supplements for disease prevention. The study found the longer people took the supplements, the more they increased their risk for lung cancer.19 Another study, focusing on the mineral selenium, found that people deficient in selenium benefited by supplementation. However, an increased rate of lung cancer occurred in people taking selenium who were not deficient.20 Use of antioxidant nutrient supplementation (i.e., Vitamin C, Vitamin E, Selenium, and others) are not recommended during radiation therapy or alkylating chemotherapies. The Academy of Nutrition and Dietetics Evidence Analysis library has graded and compared the nutrition research and is not currently recommending the use of any high-dose oral antioxidants at this time for cancer prevention nor during cancer treatment.21
Studies are currently underway to evaluate the impact of omega-3 fatty acids (fish oils) and physical activity as an intervention useful for interrupting the pre-cachexia syndrome, through their anti-inflammatory effects. Omega-3 fatty acid supplementation may improve global quality of life.15 Omega-3 oils can be found in fish such as salmon, halibut, fresh tuna, as well as flaxseed and walnuts.22-23
Bioactive compounds of interest being studied in relation to lung cancer include: Fish oil; tea (Camellia sinesis); Isothiocyanates and Indole-3-Carbinol (present in cruciferous vegetables such as cabbage, broccoli, brussels sprouts, kale and cauliflower); Genistein isoflavones (present in soybean); Curcumin (Curcuma longa); pomegranate polyphenols (Punica granatum); Fisetin flavonoid (present in strawberry, persimmon, grape, apple, cucumber and onion).24 If tolerated, it is appropriate to include these nutrients into a healthy diet. The best approach for nutrient supplementation should be individualized to each person’s background, genetic profile, lab tests, and cancer risk. Blood tests can be done to assess current levels of nutrients and potential advisability of supplementation. Recommendations about supplements may be discussed with the physician or Oncology Registered Dietitian/Nutritionist.15, 20
Managing Side Effects and Complications
Early identification and active intervention for side effects are important to protect quality of life. A significant component of cancer treatment support is geared to manage symptoms and side effects. Effective use of medication may facilitate symptom control and side effect management. The patient should routinely speak with the health care team members about medicines that may help control symptoms. Nutritional intervention may focus on lifestyle changes and behavior modification that address symptoms or side effects.11, 15
Anorexia and Early Satiety
Some patients with lung cancer may experience anorexia, also called loss of appetite. People with anorexia may describe their lack of appetite as “searching for foods that interest the taste buds” or “not being able to find anything to eat that sounds good to eat.” How does a patient eat if there is no sensation of appetite or feeling of hunger? Anorexia must be addressed because although the patient may not feel a natural appetite, the body experiences hunger, which may reveal as weakness, fatigue, excessive sleeping, and/or inability to concentrate.
Early satiety is often described as “feeling full after only a few bites”. In severe cases, people may state that they “would rather spit food out than swallow it” or “the food balls up in the mouth, and they just can’t swallow it.”
This starvation cycle can be interrupted purposefully. One well-tolerated approach is to transition from several large meals each day to smaller, more frequent meals and snacks. By eating and drinking frequently, creating a schedule for meals and snack times (even small portions count), can provide fuel adequate to improve weakness and fatigue. The anorectic patient should consciously think about eating as a way to provide vital nourishment to the muscles and immune system. In other words, “don’t wait to feel hungry—eat because it is time to eat.” If anorexia is severe, appetite stimulant medications may be considered.
Instead of asking the patient, “Are you hungry?” or “What do you want to eat?” try asking “What could you eat (or drink) right now? Or “what can you tolerate right now?” Patients should consider eating and drinking more frequently, such as every 2 to 3 hours during the day. Or, if eating is impossible, a patient may consider getting most of their nutrition from beverages. Many people experiencing anorexia for solid food still feel thirsty and can use nutritious beverages to provide calories, protein, as well as fluid. For example: 2 ounces of a milkshake taken each hour provides at least 1500 calories over a day. Some patients use a kitchen timer, cell phone alarm, or watch to cue themselves to eat and drink.
Patients who do not have much appetite, may be unable to eat the same food repeatedly or tolerate leftovers. Therefore, it is advisable to make small batches and rotate through tolerated menu items. Consider keeping a record of flavor profiles that taste good (for example: fruit flavors). These taste preferences may change based on the day of the treatment cycle, fatigue, or other factors. Creating a list of tolerated foods reassures the patient that some foods are acceptable and appealing may help stimulate ideas for other menu options.
Anorectic patients may be unable to eat the same food repeatedly or tolerate leftovers. Therefore, it is advisable to make small batches and rotate through tolerated menu items. Food may be served to the patient frequently, almost as a “surprise”. Consider keeping a record of foods and beverages that taste good or sometimes are tolerable, which may depend on the day of the treatment cycle, fatigue, or other factors. If the food does not taste good, the patient should try another type of food. Creating a list of tolerated foods reassures the patient that some foods are acceptable, and appealing may help stimulate ideas for other menu options.
Taste Changes
Taste alterations may be the side effect of cancer itself, the chemotherapy regimen, infection, or certain medications. Most taste changes develop and dissipate depending on the treatment cycle. Taste changes may negatively affect appetite, patient’s describe food flavor resembles; “flat taste” or is like “cardboard” , a metallic , food is super-salty , or “sickly sweet”. It is important to address the particular taste alteration, as described below(Table 4).22, 25-26
Table 4. Specific Suggestions for Managing Taste Changes in Patients with Lung Cancer25-26
Nausea and Vomiting
Nausea and vomiting are common side effects of many chemotherapy regimens. Most cancer centers use medication routinely to minimize nausea or vomiting. It may be helpful to maintain a record each day of a treatment cycle that nausea occurs, including the time of day and factors that influence nausea. Distinguish and note what triggers nausea or queasiness to help the health care team identify whether nausea is anticipatory, acute, delayed, or breakthrough. Each of these types of nausea may be treated differently with medication and behavioral strategies (Table 5).25, 27
Table 5. Specific Suggestions for Managing Nausea and Vomiting in Patients with Lung Cancer25, 27

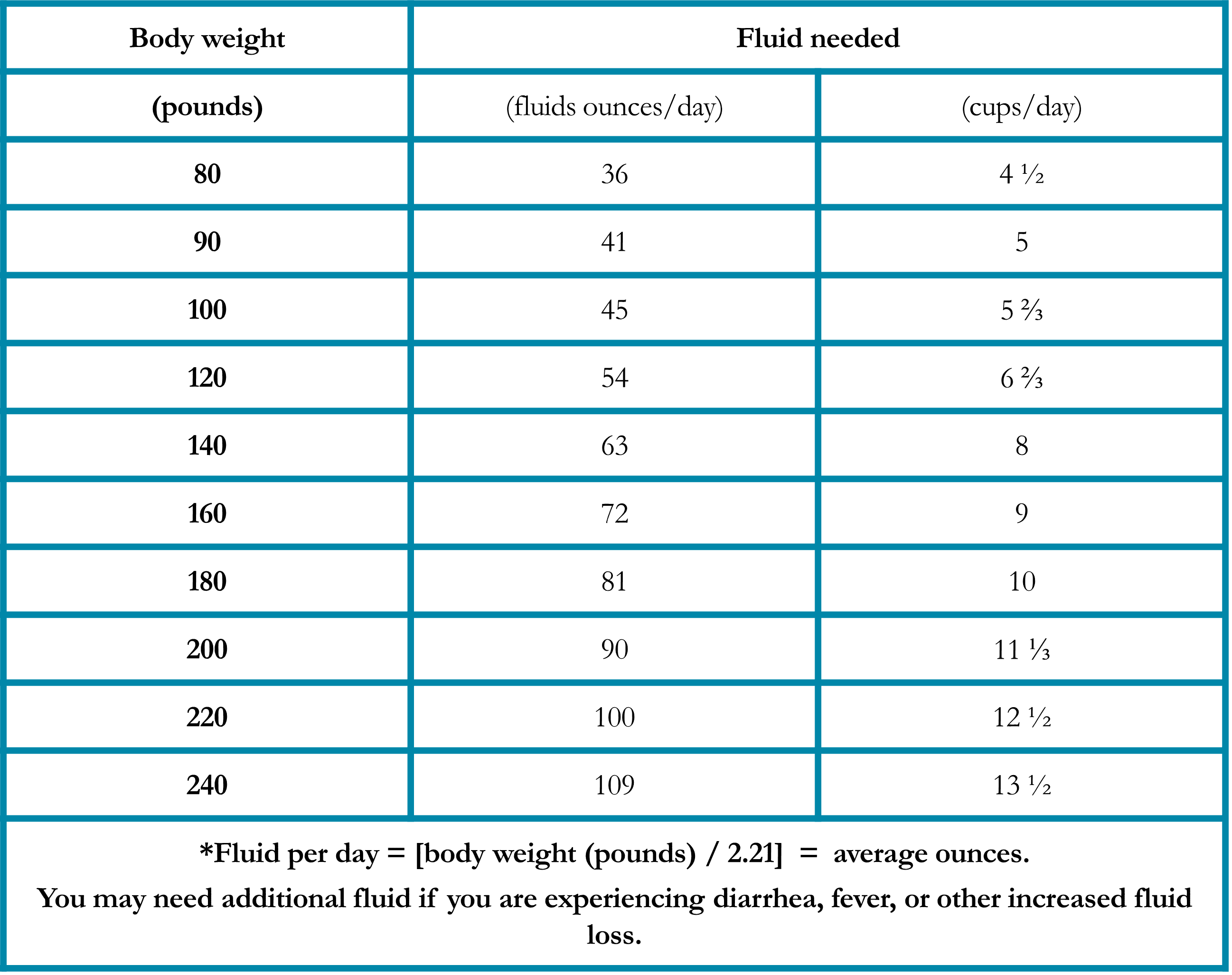
Sore Mouth and Throat
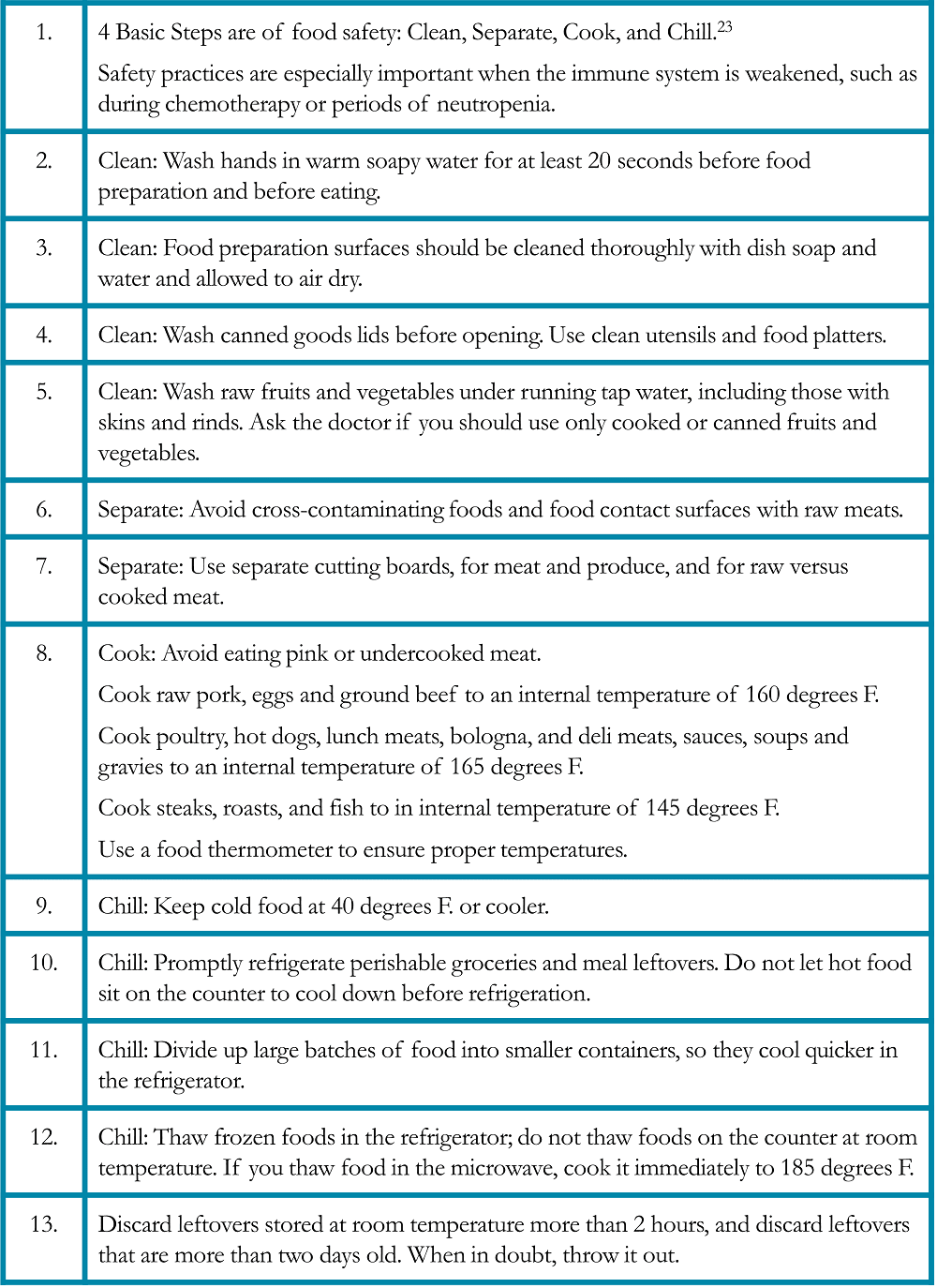
Mucositis is a painful inflammation and ulceration of the mucous membranes of the mouth and digestive tract that may be a complication of chemotherapy or radiation therapy. Oral mucositis (“mouth sores”) may cause difficulties with eating, including chewing solid food and drinking hot or acidic beverages. Radiation esophagitis is an inflammation of the esophagus after radiation therapy that may cause painful swallowing. Nutritional modifications help minimize symptoms and nutritional deficiencies resulting from these conditions (Table 6).25, 27 Foods that are recommended if diarrhea is occurring due to the side effects of immunotherapy (Table 7).25, 27 Fatigue and food safety are issues that warrant special considerations (Tables 8 and 9).28-29
Table 6. Specific Suggestions for Managing Mucositis and Radiation Esophagitis in Patients with Lung Cancer25, 27
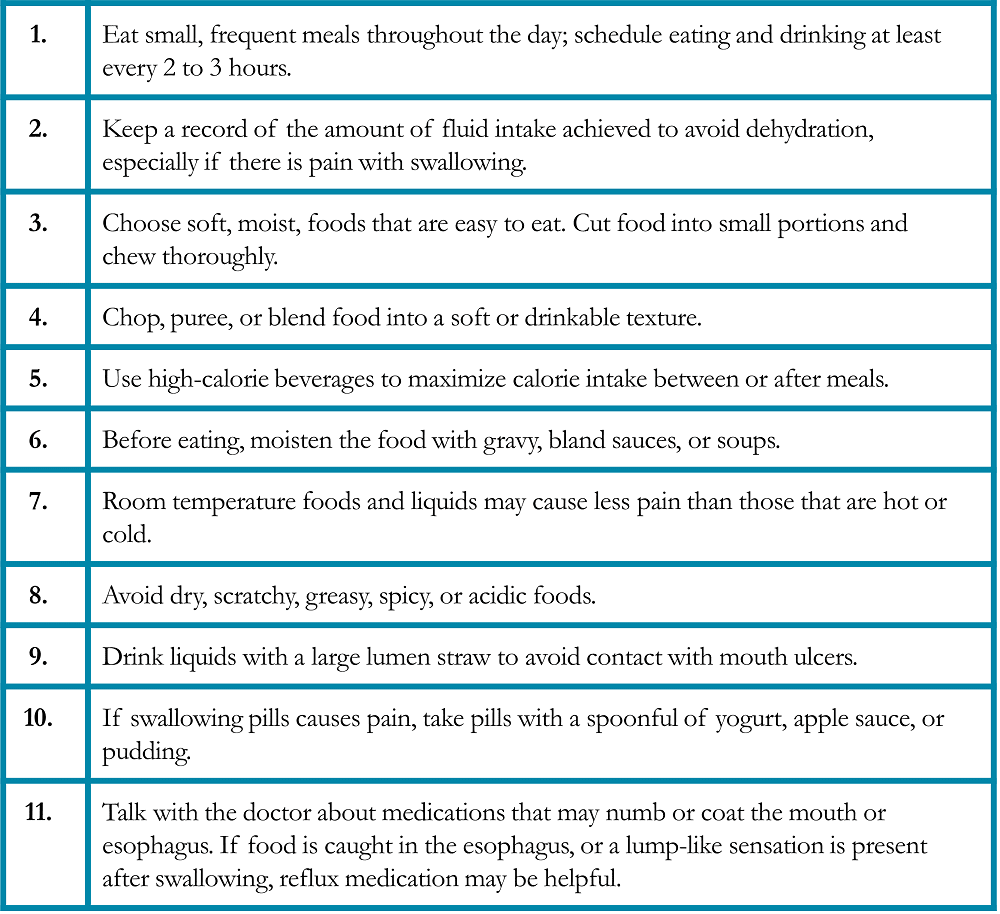
Table 7. Specific Suggestions for Managing Diarrhea in Patients with Lung Cancer25, 27
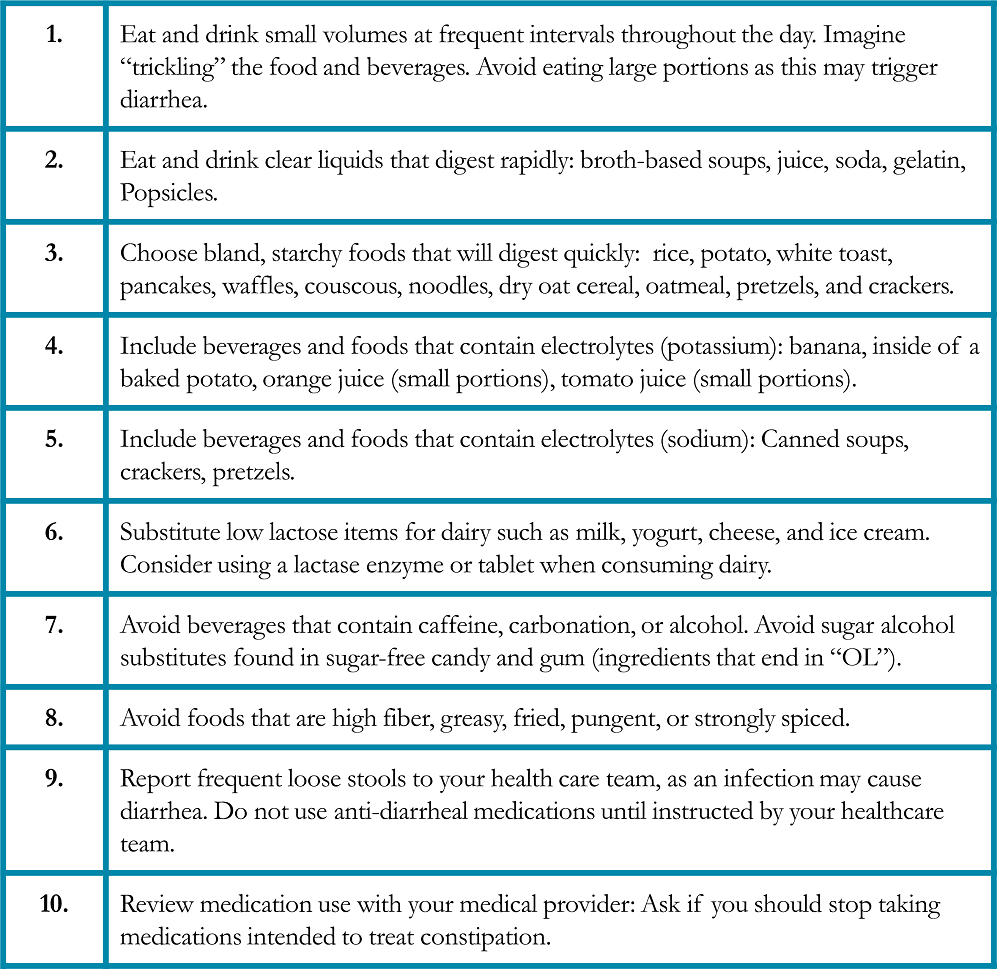
Table 8. Specific Suggestions for Managing Fatigue in Patients with Lung Cancer28-29
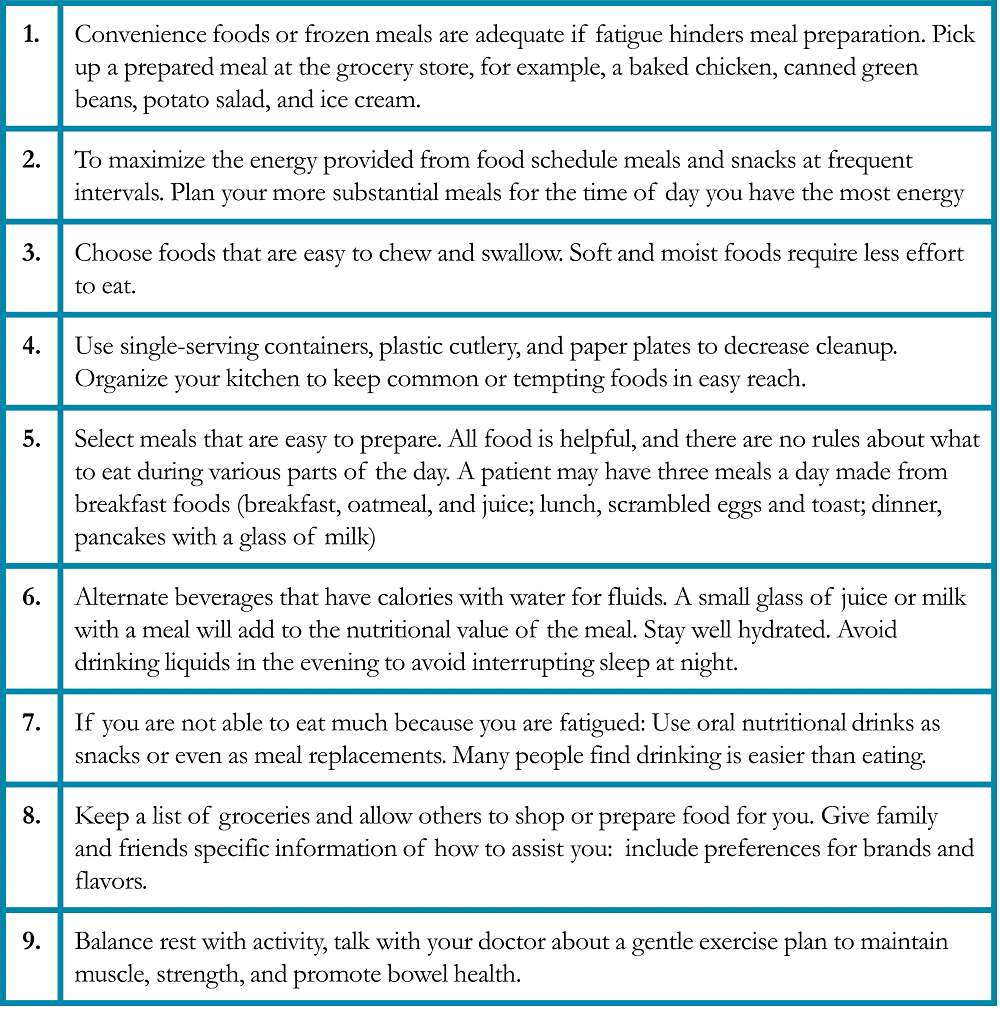
Table 9. Food Safety Suggestions for Patients with Lung Cancer28-29
Example menu if the patient has fatigue:
| Breakfast: | Instant oatmeal made with whole milk, juice, coffee with cream |
| Snack: | ¼ cup of trail mix, 6 oz. yogurt |
| Lunch: | 8 oz. can of cream soup, peanut butter and jelly sandwich, potato chips, instant iced tea |
| Snack: | Ice cream bar |
| Dinner: | Baked chicken (already prepared at a grocery store), salad mix (bagged), instant mashed potato, gravy (out of a jar), green beans (canned), a glass of chocolate milk |
| Snack: | Graham crackers, vanilla pudding (single-serve container) |

Resources for Treatment and Survivorship
The National Cancer Institute (NCI) offers a comprehensive, free resource to patients undergoing cancer treatment regarding nutrition: Eating Hints: Before, During and After Cancer Treatment.25 It can be accessed online, and downloaded or printed at: https://www.cancer.gov/publications/patient-education/eating-hints It is also available in Spanish. Other resources are available on the website of the National Cancer Institute www.cancer.gov.
The American Cancer Society (ACS) offers helpful information: Nutrition for People with Cancer: Survivorship during and after cancer treatment and Living well during treatment.29 It is available online at https://www.cancer.org/treatment/survivorship-during-and-after-treatment.html Support information is also available in Spanish as well as other languages. Other resources are available on the website of the American Cancer Society www.cancer.org.
The American Institute for Cancer Research (AICR) offers a comprehensive guide for nutrition, physical activity, weight management, and cancer prevention information. They also provide recommendations for cancer survivors. The 3rd Expert Report Diet, Nutrition, Physical Activity, and Cancer: A global perspective includes reviews of thousands of nutrition and cancer studies, to help develop public policy and personal prevention recommendations.30 In addition, the AICR routinely updates recommendations for each cancer type, reviewing the most recent research, and then combining it with previously reviewed data. The website www.aicr.org also offers updates on new research as it occurs, recipes and links to reputable resources.
References
- National Comprehensive Cancer Network, 2021. www.nccn.com. Accessed 6/14/21.
- Ross PJ, Ashley S, Norton A, et al. Do patients with weight loss have a worse outcome when undergoing chemotherapy for lung cancers? Br J Cancer. 2004; 90(10): 1905-1911.
- Morel H., Raynard B., d’Arlhac M, et al. Prediagnosis weight loss, a stronger factor than BMI, to predict survival in patients with lung cancer. Lung Cancer. 2018; 126: 55-63.
- Dewys WD, Begg C, Lavin PT, et al. Prognostic effect of weight loss prior to chemotherapy in cancer patients. Eastern Cooperative Oncology Group. Am J Med. 1980;69:491-7.
- Wang, J., Xu, H., Zhou, S., et al. Body mass index and mortality in lung cancer patients: A systematic review and meta-analysis. Eur J Clin Nutr. 2017;72(1):4-17.
- Fearon K, Arends J, Baracos V. Understanding the mechanisms and treatment options of cachexia. Nature Reviews Clinical Oncology. 2012;10(2):90-99.
- Preventing the loss of muscle mass in patients with involuntary weight loss. J Support Oncol. 2006;4(2):90-2.
- Fearon K Cancer cachexia and fat-muscle physiology. N Engl J Med, 2011;365(6):565-567.
- Adams LA, Shepard N, Caruso RA, et al. Putting evidence into practice: evidence-based interventions to prevent and manage anorexia. Clin J Oncol Nurs. 2009;13(1):95-102.
- Bauer JD, Capra S. Nutrition intervention improves outcomes in patients with cancer cachexia receiving chemotherapy – a pilot study. Support Care Cancer. 2005;13:270-4.
- Tanaka, N. Takeda K., et al. Early intensive nutrition intervention with dietary counseling and oral nutrition supplement prevents weight loss in patients with advanced lung cancer receiving chemotherapy: A clinical prospective study. Yonago Acta Medical. 2018;61:204-212.
- Aversa Z., Costelli P, Muscaritoli M. Cancer-induced muscle wasting: Latest findings in prevention and treatment. Therapeutic Advances in Medical Oncology. 2017;9(5)369-382.
- Staal-van den Brekel AJ, Dentener M, Schols A, et al. Increased resting energy expenditure and weight loss are related to a systemic inflammatory response in lung cancer patients. Journal of Clinical Oncology. 1995;13(10) 2600-2605.
- Kiss, N., Isenring, L. Academy of Nutrition and Dietetics. Oncology Nutrition for Clinical Practice (2nd Edition): Chapter 22, Medical Nutrition Therapy for Lung Cancer. 2021; 486-497.
- Atlan, P., Bayar, M., Lanoy, E., et al. Factors which modulate the rates of skeletal muscle mass loss in non-small cell lung cancer patients: A pilot study. Supportive Care Cancer. 2017;25(11):3365-3373.
- Rhoda KM, Porter MJ, Quintini C. Fluid and electrolyte management: putting a plan in motion. JPEN J Parenter Enteral Nutr. 2011;35(6):675-85.
- McCarthy D, Weihofen D. The effect of nutritional supplements on food intake in patients undergoing radiotherapy. Oncol Nurs Forum. 1999;26(5):897-900.
- Pathak AK, Bhutani M, Guleria R, et al. Chemotherapy alone vs. chemotherapy plus high dose multiple antioxidants in patients with advanced non-small cell lung cancer. J Am Coll Nutr. 2005;24(1):16-21.
- Satia JA, Littman A, Slatore CG, et al. Long-term use of beta-carotene, retinol, lycopene, and lutein supplements and lung cancer risk: results from the VITamins And Lifestyle (VITAL) study. Am J Epidemiol. 2009;169:815-28.
- Harvie, M. Nutritional supplements and cancer: Potential benefits and proven harms. 2014 ASCO Educational Book. e478-e486.
- Recommendation’s summary: Oncology (Onc) lung cancer: chemotherapy and use of antioxidant vitamins C, E and beta-carotene oral supplements. ADA Evidence Analysis Library. http://www.adaevidencelibrary.com/template.cfm?template=guide_summary&key=1836&highlight=Lung%20cancer&home=1 Accessed 4/14/21.
- Payne C, Larkin PJ, McIlfatrick S, et al. Exercise and nutrition interventions in advanced lung cancer: A systematic review. Curr Oncol. 2013; 20(4): e321-e337.
- Girolamo F, Situlin R, Mazzucco S, et al. Omega-3 fatty acids and protein metabolism: Enhancement of anabolic interventions for sarcopenia. Co-clinical Nutrition. 2014;17(2):145-150.
- Khan N., Mukhtar H. Dietary agents for prevention and treatment of lung cancer. Cancer Letters. 2015; 359(2):155-164.
- National Cancer Institute. Eating Hints: Before, During, and After Cancer Treatment. https://www.cancer.gov/publications/patient-education/eating-hints US Department of Health and Human Services, National Institutes of Health. NIH Publication. Accessed 6/14/21.
- Turcott, J.G., Juarez-Hernandez E., et al. Baseline dysgeusia in chemotherapy -naïve non-small cell lung cancer patients: Association with nutrition and quality of life. Nutrition and Cancer. 72:2, 194-201,
- Levin, R. Academy of Nutrition and Dietetics. Oncology Nutrition for Clinical Practice (2nd Edition): Chapter 11, Managing nutrition impact symptoms of cancer treatment. 2021; 194-217.
- United States Department of Agriculture. Kitchen Companion: Your safe food handbook. https://www.fsis.usda.gov/sites/default/files/media_file/2020-12/Kitchen-Companion.pdf. Accessed 6/14/21.
- American Cancer Society. Survivorship: During and after treatment. https://www.cancer.org/treatment/survivorship-during-and-after-treatment.html Accessed 6/14/21.
- American Institute for Cancer Research. Diet, Nutrition, Physical Activity and Cancer: a Global Perspective. 2018. https://www.wcrf.org/dietandcancer/contents. Accessed: 6/14/21